About Us
Executive Editor:Publishing house "Academy of Natural History"
Editorial Board:
Asgarov S. (Azerbaijan), Alakbarov M. (Azerbaijan), Aliev Z. (Azerbaijan), Babayev N. (Uzbekistan), Chiladze G. (Georgia), Datskovsky I. (Israel), Garbuz I. (Moldova), Gleizer S. (Germany), Ershina A. (Kazakhstan), Kobzev D. (Switzerland), Kohl O. (Germany), Ktshanyan M. (Armenia), Lande D. (Ukraine), Ledvanov M. (Russia), Makats V. (Ukraine), Miletic L. (Serbia), Moskovkin V. (Ukraine), Murzagaliyeva A. (Kazakhstan), Novikov A. (Ukraine), Rahimov R. (Uzbekistan), Romanchuk A. (Ukraine), Shamshiev B. (Kyrgyzstan), Usheva M. (Bulgaria), Vasileva M. (Bulgar).
Engineering
In our opinion, the use of water-activated galvanic cells for these purposes has several advantages:
Low cost and simplicity of design;
Possibility of multiple usage;
Safety in manufacture and disposal of the electrolyte.
Recycling water activated elements is not difficult, as fresh or sea water is used as the electrolyte, operating parameters being sufficiently high. Operation begins upon filling with water and does not require highly skilled maintenance.
A distinctive feature of the galvanic cell described in [3,4] is the presence of electroactivated electrolyte containing a definite concentration of NaCl, development of effective surface area of the cathode by a permanent magnet as well as a magnetic field acting on the electrolyte.
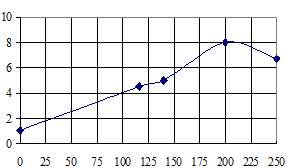
The results of the studies related to the determining the effect of the magnetic induction of permanent magnets on the parameters of a water-activated galvanic cell and which were carried out with using permanent magnets having different values of magnetic induction: В = 116; 140; 200; 250 mT (at a load of 100 ohms for 24 hours) are shown in Fig. 1. For the development of effective surface area of the cathode galvanized iron shavings were used.
 The
analysis of current strength and voltage dependence on the space between the
electrodes has led to the conclusion that the most efficient cross-section of
the electrolyte between the electrodes of the given galvanic cell is the
cross-section is in the interval of 3-5 mm.
The
analysis of current strength and voltage dependence on the space between the
electrodes has led to the conclusion that the most efficient cross-section of
the electrolyte between the electrodes of the given galvanic cell is the
cross-section is in the interval of 3-5 mm.
![]()
Fig. 1. Changes in the current strength of the galvanic cell at a different magnetic induction
|
Statistic data obtained during the experimental studies allowed to create a mathematical model of dependence of the cell current density on the magnetic field and the porosity coefficient of the electrode surfaces.
To obtain a mathematical model of the current density dependence of the cell with the developed magnet active surface of the electrodes on the magnetic induction and porosity coefficient of the electrodes a statistical analysis of the experimental data based on the use of methods of dispersion-regression analysis was carried out (Table 1).
Regression equation coefficients significance was determined by Student t-test. The actual value of the t-test was determined using the STATITIC 5.5. program.
Table 1. Experimental studies data
|
Porocity coefficient of the active electrode surface |
Magnetic induction, mT |
Current density |
|
6,0-8,2 |
0,15-0,2 |
2,1-4,8 |
Nonlinear regression equation of the second order obtained by using PC processed data has the form:
![]()
where j - current density of the galvanic cell A/m2; Cp - porosity coefficient of the active surface; B- permanent-magnet magnetic induction, T.
Equation adequacy has been verified using F-Fisher criterion, which is the ratio of dispersions.
F=12,46>4,28
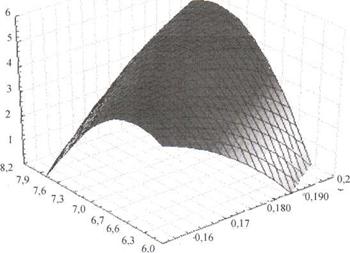
|
|||
|
|||
Fig. 2. Cal model of the dependence of water-activated galvanic cell current density on the magnetic induction and anode active surface development coefficient.
According to the received surface response shown in Fig. 2, the current density of the cell increases with the porosity ratio of the electrode active working surface and magnetic induction value increase.
Primary cells; patentee FSEIHPE Stavropol State Agrarian University. -
2007122965/22; appl. 18.06.2007; publ. 10.11.2007, Bull. Number 31.
2. Pat. Number 2343570 Russian Federation MPK8 N01M 6/32, N01M 6/34.
Primary cells; patentee FSEIHPE Stavropol State Agrarian University. -
2009122965/22; appl. 22.03.2008; publ. 10.11.2009, Bull. Number 31.
3. Kolesnikov G.Yu. Using a permanent magnetic field for increasing the capacity of a water-activated electrochemical cell / / Collection of scientific papers based on scientific and practical conference of the electricity department. Stavropol Agrus. 2009 - S. 43-46.
4. Kolesnikov G.Yu., Titovskiy D.L. Influence of the magnetic field on the energy capacity of water-activated electrochemical cells with developed electrode surfaces //Fundamental research. M.: Fundamental research. 2013 - pp. 33-36.
Kolesnikov G.Yu., Titovskiy D.L. MAGNETIC WATER ACTIVATED GALVANIC CELL WITH DEVELOPED ELECTRODE SURFACE. International Journal Of Applied And Fundamental Research. – 2014. – № 2 –
URL: www.science-sd.com/457-24640 (07.03.2026).










 PDF
PDF